

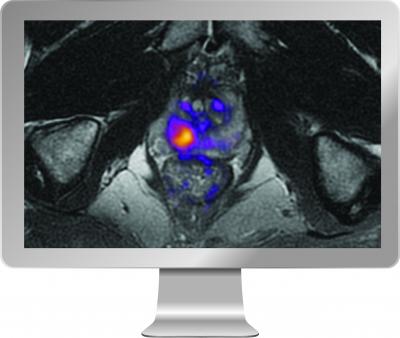
SAN DIEGO, Nov. 26, 2019 /PRNewswire/ -- HealthLytix (www.healthlytix.com), a healthcare technology company developing new advances in genetics and medical imaging to improve screening and early detection of cancer and neurodegenerative diseases, announced today that it has received 510(k) clearance from the U.S. Food and Drug Administration for its breakthrough prostate imaging software, RSI-MRI+.
RSI-MRI+ utilizes a patented advanced diffusion MRI technique called Restriction Spectrum Imaging (RSI) to help improve early detection and diagnosis of prostate cancer by clinicians. It is the first FDA-cleared imaging software to use artificial intelligence and an innovative tissue microstructure model to increase the visibility of restricted water in the body's tissue.
Many cancers, including prostate cancer, are highly cellular tissues that trap or restrict water. A limitation of conventional diffusion MRI is that it doesn't directly measure restricted water, but instead relies on a simpler measure known as the apparent diffusion coefficient (ADC). While the ADC is an important biomarker, it's affected by a number of confounding factors that can negatively impact its sensitivity and specificity to cancer, which can result in missed or inaccurate diagnoses.
"A key innovation of RSI-MRI+ is that it can better characterize the complexity of water diffusion in cancerous tissue, resulting in better performance," said Nathan White, PhD, CEO of HealthLytix and co-inventor of RSI. "With early-stage aggressive cancer, we need more sophisticated approaches to separate what's important from what's not important. In cancer, that means separating restricted diffusion from other sources of water diffusion that are less relevant."
A number of peer-reviewed articles from multiple clinical sites have shown the improved sensitivity and specificity of the RSI technique compared to conventional diffusion MRI for cancer imaging. In total, more than 20 peer-reviewed articles have been published about RSI across multiple body regions, including prostate, brain, and breast. RSI-MRI+ is currently cleared for prostate. HealthLytix plans to seek FDA clearance for additional body regions in the future.
HealthLytix is debuting RSI-MRI+ during the Radiological Society of North America (RSNA) annual meeting taking place Dec. 1-6 in Chicago. RSNA attendees interested in learning more about RSI-MRI+ are invited to visit booth #3672 in the South Hall, Level 3, of McCormick Place, or to contact Mark Wittenberg, Head of Business Development, at mark.wittenberg@healthlytix.com to set up a meeting.
About HealthLytix
At HealthLytix, we're developing cutting-edge advances in genetics and medical imaging to revolutionize disease screening and early detection so people can enjoy longer, healthier lives while reducing healthcare costs. Learn more about our personalized genetic tests and our advanced imaging solutions at www.healthlytix.com.